CE Singclean FLU A/B Antigen Test Kit (Colloidal Gold)
CE
INTENDED USE
FLU A/B Antigen Test Kit (Colloidal Gold) is used for in vitro qualitative detection of influenza A/B antigen in human nasopharyngeal swab samples.
PACK FORMATS
1 Test/Box
10 Tests/Box
20 Tests/Box
50 Tests/Box
INTRODUCTION
Influenza viruses (Flu) are the pathogens that cause influenza. Influenza is an acute respiratory infection caused by influenza A, B, and C viruses. It is infectious strong, fast spread, short incubation period, high incidence. Influenza A virus often appears in epidemic form and can cause a worldwide influenza pandemic. It is widely distributed in animals and can also cause influenza epidemics in animals and cause a large number of animal deaths. Influenza B virus often causes local outbreaks and does not cause a worldwide influenza pandemic. Therefore, the detection of influenza A and B viruses has relatively great clinical findings.
PRINCIPLE
The kit uses colloidal gold immunochromatography to detect influenza A/B virus antigens. The reagent is coated with influenza A virus monoclonal antibody (test line A), influenza B virus monoclonal antibody (test line B) and goat anti-mouse polyclonal antibody (control line C) on a nitrocellulose membrane. The gold label pad contains another influenza A virus monoclonal antibody and influenza B virus monoclonal antibody coupled with colloidal gold. When a sample containing influenza A virus antigen is added to the sample well, the influenza A virus antigen will combine with the influenza A virus monoclonal antibody conjugate to form an antigen-antibody complex. The complex moves through the nitrocellulose membrane. When the complex encounters the influenza A virus monoclonal antibody on the test line A, the complex is captured and purple-red line appeared. When a sample containing influenza B virus antigen is added to the sample well, the influenza B virus antigen is the same as that of influenza A virus antigen. The influenza B virus antigen will color the test line B, a purple-red line appeared. No color band on test line A/B means negative test result.
The test kit contains a quality control line (control line C). A purple-red line appears on the control line regardless of the presence or absence of Flu A/B virus antigen in the sample. If the control line is not colored, it means that the test result is invalid regardless of whether the test line is colored. The control line is the criterion for judging whether the test strip is valid.
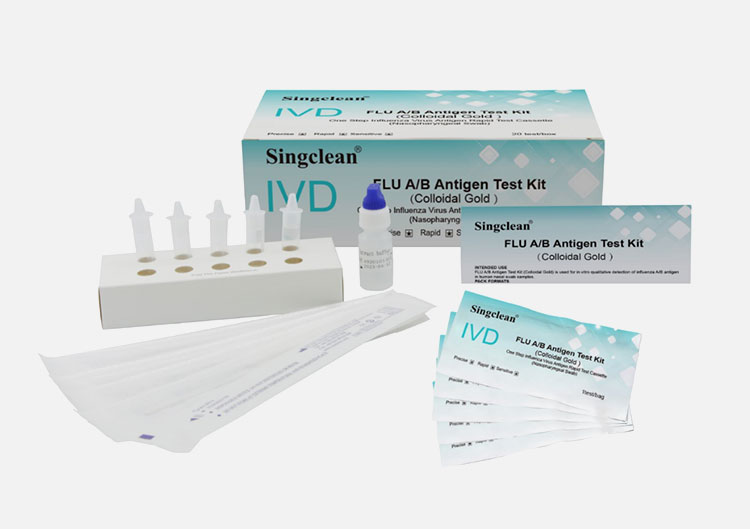
MATERIALS SUPPLIED
1. Test Cassette
2. Sampling cotton swabs (as nasopharyngeal swab)
3. Antigen extraction buffer
4. Antigen extraction tube
5. Paper workbench(The small one-test-box can be used as a workbench)
6. Instruction for use
MATERIALS REQUIRED BUT NOT PROVIDED
Timer
STORAGE AND STABILITY
The kit can be stored at 4-30°C. The test device is stable through the expiration date printed on the sealed pouch. The test device must remain in the sealed pouch until use.
Do not freeze. Do not use beyond the expiration date. After opening the sealed pouch, use the test as soon as possible within 60 minutes.
WARNINGS AND PRECAUTIONS
1. For professional In Vitro diagnostic use only. Do not use after expiration date.
2. These instructions must be strictly followed by a trained healthcare professional to achieve accurate results. All users have to read the instruction prior to performing a test.
3. Do not use it if the tube/pouch is damaged or broken.
4. Do not use if package is open or damaged.
5. Wear protective while handling specimens and wash hands thoroughly afterwards.
6. Avoid splashing or aerosol formation of specimen and buffer.
7. Clean up spills thoroughly using an appropriate disinfectant.
8. Decontaminate and dispose of all specimens, reaction kits and potentially contaminated materials (i.e. swab, extraction tube, test device) in a biohazard container as if they were infectious waste and dispose according to applicable local regulations.
9. Do not mix or interchange different specimens.
10. Do not mix reagent of different lots or those for other products.
11. Do not store the test kit in direct sunlight.
12. To avoid contamination, do not touch the head of provided swab when opening the swab pouch.
13. The provided swabs in the package should be used only for nasopharyngeal specimen collection.
14. To avoid cross-contamination, do not reuse the swabs for specimen collection.
15. Do not dilute the collected swab with any solution except for the provided extraction buffer.
16. Test is for single use only. Do not re-use under any circumstances.
17. Do not perform the test in a room with strong air flow, ie. electric fan or strong air-conditioning.
18. In case of positive samples, seek medical attention in time.
19. lf the extracting solution touches eyes and mouth, a large amount of clean water should be rinsed in time and medical treatment should be sought if needed.
20. In case of any serious incident related to the device, it shall be reported to the manufacturer, distributor, EU authorised representative, notified body and the competent authority.
SPECIMEN COLLECTION
1. FLU A/B Antigen Test kit(Colloidal Gold)can be performed using nasopharyngeal swab.
2. Testing should be performed immediately after specimen collection.
3. Bring specimens to room temperature prior to testing.
4. If specimens are to be shipped, they should be packed in compliance with local regulations covering the transportation of etiologic agents.
TEST PROCEDURE
Allow test cassette, Antigen extraction buffer, specimen and/or controls to equilibrate to room temperature prior to testing.
1. Ask the patient to remove the secretions on the surface of the anterior nasal cavity, keep the head slightly tilted, and gently and slowly insert the swab through the nasal cavity to the nasopharynx, When resistance is encountered, it will reach the posterior nasopharynx, stay for a few seconds to absorb secretions, and gently rotate to remove the swab.
2. Place the antigen extraction tube on the workbench. Place the antigen extraction buffer bottle vertically downward, squeeze the bottle to make the buffer drip freely into the antigen extraction tube without touching the edge of the tube, and add 6 drops (about 200ul) to the antigen extraction tube.
3. Put the swab specimen into the antigen extraction tube pre-added with the antigen extraction buffer, and rotate the swab about 10 times while pressing the swab head against the tube wall to release the antigen in the swab, then let it stand for about 1 minute.
4. Remove the swab while squeezing the tip of the swab so that as much liquid in the swab can be discharged as possible. Dispose of used swabs in accordance with biohazard waste disposal methods.
5. Install the dripper on the antigen extraction tube and cap it tightly, and let it stand for about 1 minute.
6. Remove the test cassette from the sealed foil pouch and use it as soon as possible.
7. Place the test device on a clean and level surface.
8. Transfer 3 drops(about 100ul) of mix Liquids to the sample well of the test card (or use a pipette to add 100ul), and start the timer.
9. Wait for the test result of the reagent. The result should be read in 15 minutes. Do not interpret the result after 20 minutes.
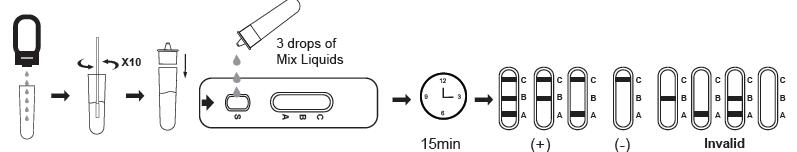
INTERPRETATION OF RESULTS
NEGATIVE:
If only the C band is present, the absence of any burgundy color in the T band indicates that no FLU A/B antigens are detected in the specimen. The result is negative.
POSITIVE
FLU A/B positive:
In addition to the presence of the C-line, If the test lines A and B appears at the same time, it means that there are both influenza A virus antigen and influenza B virus antigen in the sample, that is, the result is positive for FLU A and FLU B.
FLU A positive:
In addition to the presence of the C-line, if the test line A appears, the test indicates the presence of FLU A antigen in the sample, that is, the result is positive for FLU A.
FLU B positive:
In addition to the presence of the C-line, if the test line B appears, the test indicates the presence of FLU B antigen in the sample, that is, the result is positive for FLU B.
INVALID:
Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test with a new test cassette.
If the problem persists, discontinue using the test kit immediately and contact your local distributor.

LIMITATIONS
1. Use fresh samples whenever possible.
2. Optimal assay performance requires strictly adherence to the assay procedure described in Instruction for use. Deviations may lead to aberrant results.
3. A negative result for an individual subject indicates absence of detectable FLU A/B antigen. However, a negative test result does not preclude the possibility of exposure to or infection with FLU A/B.
4. A negative result can occur if the quantity of the FLU A/B antigen present in the specimen is below the detection limits of the assay, or failed to collect the FLU A/B antigen in the nasal cavity of the patient.
5. As with all diagnostic tests, a definitive clinical diagnosis should not be based on the result of a single test, but should only be made by the physician after all clinical and laboratory findings have been evaluated.
Latest version 8.52.04.001 B1 lssued Date: 2022.12.30